BPC-157
What The Science Actually Shows About This Healing Peptide in 2026
BPC-157 is the most-discussed healing peptide in the performance and biohacking space — and probably the most misrepresented. It has over 200 animal studies behind it, three small human pilot studies, and zero completed randomized controlled trials. Its regulatory status shifted twice in three years. And it carries a genuine theoretical cancer risk that deserves an honest look.
This guide covers what the evidence actually shows, what it doesn’t, and what the legal situation means for you right now.
Key takeaways
- Body Protection Compound-157 (BPC-157) is a synthetic 15-amino acid peptide with 207+ preclinical studies but only three small human pilot studies (~30 total subjects) and zero completed randomized controlled trials.
- BPC-157 promotes tissue repair through three primary mechanisms: VEGFR2-mediated angiogenesis, nitric oxide modulation via the Src-Caveolin-1-eNOS pathway, and FAK-paxillin cell migration signaling.
- The FDA classified BPC-157 as Category 2 in 2023, prohibiting compounding. HHS Secretary Kennedy announced reclassification to Category 1 on February 27, 2026 — but as of March 2026, the FDA has not finalized the change. Compounding remains prohibited.
- WADA added BPC-157 to its Prohibited List under S0 (Non-Approved Substances) in 2022. It is banned at all times with no Therapeutic Use Exemption available.
- Between 12% and 58% of ergo-nutritional supplements contain undeclared contaminants. Product quality is the most concrete documented safety risk for current users.
What is BPC-157?
Body Protection Compound-157 (BPC-157) is a synthetic pentadecapeptide — a chain of 15 amino acids with a molecular weight of 1,419 Daltons. Dr. Predrag Sikiric and his team at the University of Zagreb first identified it in 1993 as a fragment of a larger protein found in human gastric juice. Its amino acid sequence is Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val.
BPC-157 exists in multiple salt forms. The acetate salt is the standard formulation for most injectable preparations. The arginate salt — also marketed as Pentadeca Arginate (PDA) — bonds BPC-157 to L-arginine for improved pH stability and patent-claimed oral bioavailability. A sodium salt form also exists but is less commonly used.BPC-157 vs. PDA: PDA is not a different peptide. Pentadeca Arginate is the identical 15-amino acid sequence in a different salt formulation. Same peptide, different counter-ion. Diagen holds the patent for the arginate form.
How does BPC-157 work?
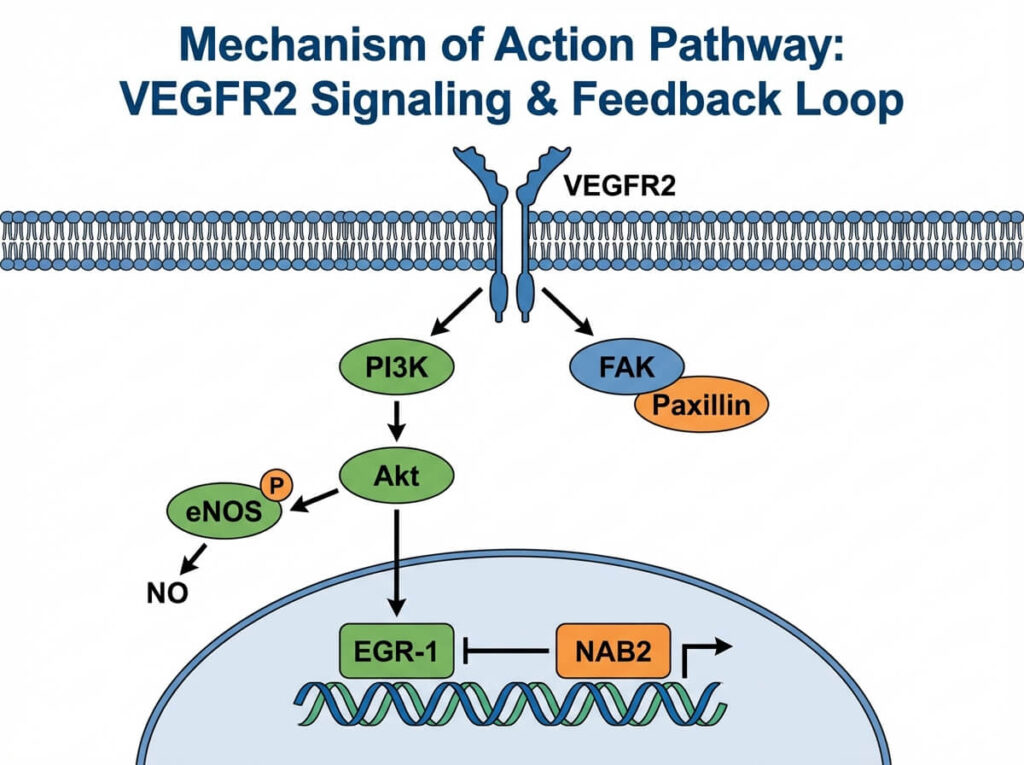
BPC-157 does not work through a single pathway. It activates at least three distinct signaling mechanisms which is part of why it shows activity across such a wide range of tissue types.
VEGFR2-Akt-eNOS angiogenesis pathway
BPC-157 activates vascular endothelial growth factor receptor 2 (VEGFR2), a primary trigger for new blood vessel formation. Chang et al. (2014) demonstrated that BPC-157 enhances growth hormone receptor expression in tendon fibroblasts through this pathway. VEGFR2 activation then initiates an Akt-eNOS signaling cascade that produces nitric oxide (NO), which dilates blood vessels and increases blood flow to injured tissue.
New blood vessels mean more oxygen and nutrients reaching the repair site — a prerequisite for meaningful tissue healing.
Nitric oxide modulation
BPC-157 modulates the nitric oxide system bidirectionally through the Src-Caveolin-1-eNOS signaling pathway. It can both upregulate and normalize NO production depending on tissue conditions. This bidirectional capacity is unusual among peptides and may explain BPC-157’s observed activity across diverse injury types and organ systems.
FAK-paxillin cell migration
BPC-157 activates the focal adhesion kinase (FAK)-paxillin signaling pathway, which governs cell attachment and migration. FAK-paxillin signaling enables fibroblasts and endothelial cells to move into damaged tissue areas and anchor to the extracellular matrix. This mechanism complements the angiogenesis pathway: BPC-157 builds new blood vessels and ensures repair cells can populate them.
EGR-1/NAB2 self-limiting feedback
BPC-157 upregulates Early Growth Response protein 1 (EGR-1), a transcription factor involved in tissue repair. EGR-1 in turn activates NGFI-A Binding Protein 2 (NAB2), which acts as a negative feedback regulator. This EGR-1/NAB2 loop may prevent excessive angiogenic signaling — a mechanism directly relevant to the cancer risk discussion covered in the safety section.
The full signaling picture:
BPC-157
→ activates VEGFR2
→ Akt-eNOS cascade → nitric oxide → vasodilation + angiogenesis
→ activates FAK-paxillin → cell migration into repair site
→ upregulates EGR-1 → NAB2 negative feedback (self-limiting)
Evidence grade: PeptideRx rates the evidence for BPC-157’s mechanism of action as Grade B: strong animal model data with plausible molecular mechanisms, but limited direct confirmation in human tissue studies.
What does the research show?
The BPC-157 evidence base spans 207+ PubMed-indexed studies published between 1993 and 2025. A 2025 systematic review by Vasireddi et al. at Case Western Reserve University screened 544 articles and included 36 studies: 35 preclinical (animal) and 1 clinical (human). Zero completed randomized controlled trials exist for any indication.
Three small human pilot studies have been published as of early 2026 — all conducted by the same research group in Florida. Total human subjects across all three trials: approximately 30.
Evidence gap: A Phase I safety trial (NCT02637284) enrolled 42 healthy volunteers in 2015 and was cancelled in 2016 with results never published.

Musculoskeletal healing
Preclinical studies consistently show BPC-157 improving outcomes in tendon, ligament, muscle, and bone injury models:
- Staresinic et al. (2003) reported improved Achilles tendon healing in rats
- Krivic et al. (2006) demonstrated accelerated tendon-to-bone repair and found BPC-157 opposed corticosteroid-induced healing impairment
- Cerovecki et al. (2010) showed improved medial collateral ligament healing in a rat transection model
- Sebecic et al. (1999) found BPC-157 promoted bone defect repair at rates comparable to autologous bone grafting
The only published human musculoskeletal study: Lee and Padgett (2021) conducted a retrospective review of 16 patients who received intra-articular BPC-157 injections for chronic knee pain. Of those 16 patients, 14 (87.5%) reported significant pain relief at 6–12 months follow-up. The study had no placebo control group, used no standardized diagnosis criteria, and represents Level IV evidence.
The Vasireddi et al. (2025) systematic review concluded that BPC-157 reduces inflammation, promotes vascularity, and improves structural and biomechanical recovery across preclinical fracture, muscle, tendon, and ligament injury models. The same review noted that in-human clinical safety remains unknown.
Gastrointestinal healing
BPC-157 originates from gastric juice and shows particular activity in GI tissue models. Preclinical studies report protection against gastric and duodenal ulcers induced by stress, ethanol, and NSAIDs — with some animal models showing favorable comparison to H2 receptor antagonists like famotidine and cimetidine. Additional animal data covers fistula healing, anastomosis improvement, and short bowel syndrome.
BPC-157 is uniquely stable in gastric acid for over 24 hours, which makes oral administration viable for GI-targeted protocols.
Lee et al. (2024) conducted a pilot study treating 12 women (ages 39–76) with moderate to severe interstitial cystitis using intravesical (bladder) BPC-157 injections of 10 mg. Ten of 12 patients reported complete symptom resolution at 6-week follow-up. The remaining two reported 80% symptom reduction. Zero adverse events were recorded. All 12 participants had previously failed pentosan polysulfate therapy, the only FDA-approved treatment for interstitial cystitis.
Neurological evidence
Animal studies report BPC-157 activity in traumatic brain injury models (Tudor et al., 2010), ischemia-reperfusion injury (Vukojevic et al., 2020), Parkinson’s disease models, serotonergic modulation, and peripheral nerve regeneration. All neurological evidence is from animal models exclusively.
Lee and Burgess (2025) conducted the most recent human study: a pilot safety trial in two healthy adults (one 58-year-old male, one 68-year-old female) who received intravenous BPC-157 infusions of 10 mg on day 1 and 20 mg on day 2. No adverse events occurred. Plasma BPC-157 concentrations returned to baseline within 24 hours.
Evidence grade: PeptideRx rates the overall clinical evidence for BPC-157 as Grade C: primarily animal and in vitro data, with limited and methodologically weak human clinical evidence.
Who uses BPC-157 and why?
BPC-157 has gained traction across five main user groups.
Recreational athletes and gym-goers represent the largest segment. CrossFit participants, weightlifters, and weekend athletes seek BPC-157 for injury recovery when conventional approaches — cortisone injections, physical therapy, surgical wait-and-see — have been slow or unsatisfactory. Common injury targets include rotator cuff tears, Achilles tendinopathy, plantar fasciitis, and herniated discs.
Biohackers and cognitive optimizers, often from tech and executive demographics, evaluate BPC-157 as part of broader optimization protocols. This group primarily discovers BPC-157 through podcasts like Huberman Lab and Reddit communities including r/Peptides and r/Nootropics.
GI and functional medicine patients explore BPC-157 for gut-related conditions including inflammatory bowel disease, chronic gastritis, and post-antibiotic gut repair.
Anti-aging and longevity-focused users investigate BPC-157 for tissue quality, cellular repair, and longevity protocols.
Patients seeking alternatives who have exhausted conventional options pursue BPC-157 for conditions where approved treatments have failed — as in the interstitial cystitis pilot study.
Google search interest for “BPC-157” reached an all-time high in June 2024. Andrew Huberman has discussed personal use and cited 300–500 mcg subcutaneously two to three times per week while cautioning about cancer risk. HHS Secretary Kennedy’s February 27, 2026 appearance on the Joe Rogan Experience (Episode #2461) further elevated public interest.
Military personnel: The Department of Defense’s Operation Supplement Safety (OPSS) program prohibits BPC-157 for all service members. Military personnel face disciplinary action for using BPC-157 regardless of how it is sourced.
Administration routes
BPC-157 can be administered through five routes, each with different bioavailability profiles and condition suitability.

| Route | Bioavailability | Best use | Evidence tier |
|---|---|---|---|
| Subcutaneous injection | Near-complete | Musculoskeletal injuries, systemic recovery | Most studied |
| Intramuscular injection | Near-complete | Deep muscle injuries | Moderate; requires professional supervision |
| Oral (acetate salt) | ~3% | GI-targeted conditions | Supported by GI stability data |
| Oral (arginate/PDA) | Up to ~90% (one rat study; no human data) | GI-targeted conditions; improved stability | Limited |
| Nasal | Uncharacterized | Hypothesized neurological applications | Minimal published data |
| Topical | Uncharacterized | Wound healing (one animal study) | Minimal published data |
Subcutaneous injection is the most studied and most commonly used route. Practitioners typically inject near the injury site for localized conditions using 29–31 gauge insulin syringes at a 45-degree angle.
Oral administration is viable specifically because BPC-157 maintains integrity in gastric acid for over 24 hours — a property unusual among peptides. The acetate salt has approximately 3% oral bioavailability. The arginate salt (PDA) carries patent-claimed bioavailability up to approximately 90%, based on a single rat study with no human confirmation. Oral is generally preferred for GI-targeted protocols.
Intramuscular injection targets deeper tissue and is sometimes preferred for muscle injuries. IM administration typically requires professional supervision.
Dosing protocols
Important: No FDA-approved human dosing exists for BPC-157. All protocols below are extrapolated from animal study doses and practitioner or anecdotal reports. Vasireddi et al. (2025) explicitly stated that current evidence prevents proposing evidence-based practice use guidelines. Consult a licensed physician before using any peptide.
Preclinical studies used 10 mcg/kg body weight as a standard reference dose. Safety studies tested ranges from 6 mcg/kg up to 20 mg/kg without achieving a lethal dose. Weight-based human extrapolation places the commonly cited range at approximately 1–2 mcg/kg per day.
Commonly reported protocols by goal
| Goal | Dose | Route | Duration |
|---|---|---|---|
| Acute injury recovery | 500–750 mcg/day | SubQ near injury site | 4–6 weeks |
| Gut healing | 250–500 mcg twice daily on empty stomach | Oral (arginate preferred) | 4–6 weeks |
| General maintenance | 250 mcg/day | Oral | 4 weeks on, 2 weeks off |
| Neurological support | 250–500 mcg/day | SubQ or nasal | Not validated |
BPC-157’s pharmacological half-life is under 30 minutes via IM or IV routes. Tissue repair effects accumulate over repeated dosing cycles. Cycle length guidance suggests 4–8 weeks of active use followed by 2–4 weeks off, though no clinical data supports specific cycling schedules.
Reconstitution and practical guide
BPC-157 for injection is typically sold as lyophilized (freeze-dried) powder in 5 mg or 10 mg vials. Reconstitution requires bacteriostatic water (BAC water) and basic sterile technique.
Reconstitution math for a 5 mg vial
| BAC water added | Concentration | Volume per 250 mcg dose | Volume per 500 mcg dose |
|---|---|---|---|
| 1 mL | 5,000 mcg/mL | 0.05 mL (5 units) | 0.10 mL (10 units) |
| 2 mL | 2,500 mcg/mL | 0.10 mL (10 units) | 0.20 mL (20 units) |
| 5 mL | 1,000 mcg/mL | 0.25 mL (25 units) | 0.50 mL (50 units) |
Step-by-step reconstitution
- Allow the lyophilized vial to reach room temperature (~15 minutes)
- Wash hands and wear nitrile gloves
- Swab both vial stoppers with alcohol pads
- Draw the desired volume of BAC water into a syringe
- Insert the needle into the BPC-157 vial and release water slowly along the inside wall — do not spray directly onto the powder
- Swirl gently until dissolved — never shake
- Label the vial with date, concentration, and contents; refrigerate immediately
Storage
| State | Temperature | Duration |
|---|---|---|
| Lyophilized (unreconstituted) | Room temp (short-term) | ~3 weeks |
| Lyophilized (long-term) | −18°C or below | Extended |
| Reconstituted | 2–8°C (refrigerated) | 2–4 weeks |
Never freeze reconstituted peptide solution.
Injection site mapping by condition
| Condition | Injection site |
|---|---|
| Shoulder injury | Deltoid region (SubQ) |
| Knee injury | Periarticular SubQ around the joint line |
| GI conditions | Abdominal SubQ |
| Systemic/general recovery | Abdominal fat pad (SubQ) |
| Muscle tear | Adjacent to injury site |
Use a 29–31 gauge insulin syringe. Insert at a 45-degree angle for SubQ injections. Rotate injection sites daily to prevent tissue irritation. Dispose of needles in a proper sharps container.
Stacking and combinations
BPC-157 + TB-500: the Wolverine Stack
The combination of BPC-157 and Thymosin Beta-4 fragment (TB-500) is the most widely discussed peptide stack for tissue repair.
TB-500 is a 43-amino acid peptide that regulates actin, a protein involved in cell structure and movement. TB-500 acts systemically — promoting cell migration throughout the body. BPC-157 acts locally — driving angiogenesis and tissue-specific repair at the injury site.
| Attribute | BPC-157 | TB-500 |
|---|---|---|
| Size | 15 amino acids, 1,419 Da | 43 amino acids |
| Origin | Human gastric juice | Thymus gland |
| Primary mechanism | VEGFR2 angiogenesis + NO modulation | Actin regulation + systemic cell migration |
| Action scope | Local (site-specific) | Systemic (whole body) |
| Oral stability | Stable in gastric acid >24 hours | Not orally stable |
| WADA status | Prohibited (S0) | Prohibited (S0) |
| FDA status | Category 2 (reclassification pending) | Category 2 |
The complementary mechanism provides the rationale for combining the two: BPC-157 builds new blood vessels and anchors repair cells locally; TB-500 mobilizes cells and reduces inflammation systemically.
Commonly reported protocol: BPC-157 at 250–500 mcg SubQ daily paired with TB-500 at 2–5 mg SubQ twice weekly for 4–8 weeks. Separate vials are generally preferred over pre-blended products to allow independent dose adjustment and to avoid potential cross-degradation.
Other reported combinations
BPC-157 + KPV: KPV is an anti-inflammatory tripeptide. The combination targets gut healing protocols where inflammation management complements tissue repair.
BPC-157 + GHK-Cu: GHK-Cu supports collagen synthesis and connective tissue remodeling. The pairing is sometimes called the “Hyper Recovery Stack” for skin and joint repair.
BPC-157 + CJC-1295/Ipamorelin: BPC-157 upregulates growth hormone receptors in tendon fibroblasts. Adding a growth hormone secretagogue may amplify circulating GH effects at BPC-157-sensitized receptor sites. This interaction is theoretical — no direct combination studies exist.
Data gap: Zero formal drug interaction studies exist for BPC-157 with any compound. Theoretical cautions apply when combining BPC-157 with pro-angiogenic compounds in individuals with cancer history, and with NO-pathway medications such as PDE5 inhibitors (sildenafil, tadalafil) and nitrates.
Side effects and safety
Animal safety data
Preclinical safety studies report no lethal dose for BPC-157. The LD1 was not reached at ranges up to 20 mg/kg in rodents. Four studies assessed organ-system safety in preclinical models and found no adverse effects across hepatic, renal, cardiac, and neurological parameters in 4-week repeated dosing protocols.
Human safety data
Across three published human studies, approximately 30 total subjects received BPC-157 with zero reported adverse events. Lee and Padgett (2021) administered intra-articular knee injections to 16 patients with no adverse effects. Lee et al. (2024) administered intravesical BPC-157 to 12 interstitial cystitis patients and screened for fevers, skin rash, nausea, vomiting, urinary symptoms, and dyspareunia — zero participants experienced any of these. Lee and Burgess (2025) administered IV BPC-157 up to 20 mg in two healthy adults with no adverse events and no clinically meaningful changes in vital signs, ECG, or blood panels.
A sample size of approximately 30 total subjects is insufficient to detect rare adverse events. Long-term safety data in humans does not exist.
Anecdotal side effects (unvalidated)
Online user reports — not scientifically validated — include injection-site pain and swelling, joint pain, anxiety, heart palpitations, insomnia, drowsiness, fatigue, loss of appetite, and depressive symptoms. Frequency and causality cannot be confirmed without controlled trials.
Cancer and tumorigenesis — the honest picture
BPC-157 upregulates VEGFR2, which drives angiogenesis. Tumors require blood vessel growth to expand beyond a few millimeters. BPC-157 also activates FAK-paxillin signaling, which can promote cell migration — a mechanism involved in metastasis. This creates a theoretical dual risk: promoting blood supply to dormant tumors and enabling cancer cell movement.
Against this: no carcinogenic effects have been documented in any published BPC-157 study. The EGR-1/NAB2 feedback loop may self-limit excessive angiogenic signaling. Radeljak et al. (2004) reported that BPC-157 reduced VEGF signaling in melanoma cells in vitro. No dedicated in vivo tumor biology study has been conducted.
Contraindication: BPC-157 is contraindicated for individuals with active cancer, cancer history, or family history of angiogenesis-dependent malignancies. Consult an oncologist before considering BPC-157 in any cancer-adjacent context.
Contamination and product purity
The Vasireddi et al. (2025) systematic review noted that between 12% and 58% of ergo-nutritional supplements contain undeclared and often unsafe contaminants. BPC-157 sold as a “research chemical” is not manufactured under FDA Good Manufacturing Practice (GMP) requirements. Independent testing has found products with incorrect amino acid sequences, purity levels as low as 5–75%, and contamination with heavy metals including arsenic and lead.
Product contamination is the most concrete, well-documented safety risk for current BPC-157 users.
Full contraindications list
- Active cancer or cancer history
- Pregnancy and breastfeeding (no safety data)
- Cardiovascular conditions affected by NO modulation
- Autoimmune conditions (immunogenicity risk)
- Children and adolescents (no pediatric data)
- Concurrent NO-pathway medications without physician oversight
Legal and compliance status in 2026
FDA classification
The FDA classified BPC-157 as a Category 2 bulk drug substance in 2023, citing significant safety concerns. Category 2 status prohibits licensed compounding pharmacies from using BPC-157 to prepare medications for human use under Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act. The FDA cited insufficient human safety data, immunogenicity risk, and characterization and impurity concerns.
On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced on the Joe Rogan Experience (Episode #2461) that approximately 14 of the 19 Category 2 peptides — including BPC-157 — would be reclassified to Category 1, allowing licensed compounding.
Current status as of March 2026: The FDA has not published its official updated Category list. The reclassification remains pending finalization. Compounding BPC-157 remains prohibited under federal law until formal FDA documentation is published. Verify current status at FDA.gov before making any decisions.
Category 1 reclassification does not equal FDA drug approval. BPC-157 would remain an off-label, unapproved compound requiring physician supervision and a valid prescription.
DEA status
The Drug Enforcement Administration does not schedule BPC-157. Possession is not illegal for individuals — unlike anabolic steroids. BPC-157 occupies a regulatory gap: it cannot be legally compounded or sold for human use, but individual possession carries no criminal penalty.
WADA and sports bans
WADA added BPC-157 by name to its Prohibited List under Section S0 (Non-Approved Substances), effective January 1, 2022. S0 covers any substance not approved by a governmental regulatory health authority for human therapeutic use. BPC-157 is prohibited at all times, in and out of competition, with no Therapeutic Use Exemption available.
| Organization | BPC-157 status | Year enacted |
|---|---|---|
| WADA | Prohibited (S0, named specifically) | 2022 |
| USADA | Prohibited (enforces WADA S0) | 2022 |
| NFL | Prohibited (specifically listed) | 2022 |
| UFC | Prohibited (specifically listed) | 2022 |
| NCAA | Prohibited (2026 banned substance handbook) | 2022+ |
| NBA | Prohibited (general peptide hormone ban) | Ongoing |
| NHL | Prohibited (general peptide hormone ban) | Ongoing |
| MLB | Prohibited (general peptide hormone ban) | Ongoing |
| DoD/OPSS | Prohibited (prohibited dietary supplement ingredient) | Ongoing |
BPC-157 is detectable in urine for up to 4 days via high-resolution mass spectrometry. The Lee and Burgess (2025) IV pilot study showed plasma levels return to baseline within 24 hours, but urine metabolites persist longer.
Gray market access
BPC-157 is currently sold online by vendors labeling it as a “research chemical” with disclaimers such as “not for human consumption.” These vendors operate in a legal gray area: the peptide itself is not DEA-scheduled, but selling it for human therapeutic use violates FDA regulations. Enforcement actions have included the Tailor Made Compounding LLC case, resulting in a guilty plea and $1.79 million forfeiture.
Buying guide and quality evaluation
BPC-157 is available as lyophilized powder vials (5 mg and 10 mg, most common), oral capsules (typically arginate salt), nasal spray formulations, pre-blended combination vials, and topical creams. BPC-157 is not available through Amazon, GNC, Vitamin Shoppe, or any mainstream retail supplement channels.
Price benchmarks (early 2026)
| Source type | 5 mg vial price range |
|---|---|
| Research chemical vendors | $35–$65 |
| Compounding pharmacies (when legal) | $80–$150+ |
| Oral capsules (30-day supply) | $40–$90 |
Six quality markers to verify
Third-party Certificate of Analysis (COA). A COA from an independent laboratory is the minimum quality requirement. It must be batch-specific, not generic.
HPLC purity of 97% or above. High-performance liquid chromatography testing should confirm peptide purity at 97% or higher.
Salt form explicitly stated. The label or COA should specify whether the product is BPC-157 acetate, arginate, or sodium salt.
USA-based manufacturing. Domestic manufacturing provides more accountability than overseas-only sourcing.
GMP-certified facility. Good Manufacturing Practice certification indicates standardized quality control, though GMP is not FDA-mandated for research chemicals.
Batch-specific testing. Each production batch should have its own COA. A single COA applied to all batches is a red flag.
Red flags
- No COA available, or COA provided only after purchase
- Pre-mixed liquid peptides (reconstituted peptide has limited shelf stability)
- Prices below $20 per 5 mg vial (below manufacturing cost for quality product)
- Therapeutic claims on the product label (violates FDA regulations)
- No company contact information, address, or customer service
- Overseas-only shipping with no domestic presence
Compounding pharmacy access for BPC-157 is expected to resume after the FDA formally finalizes Category 1 reclassification. Compounding pharmacy preparations offer pharmaceutical-grade quality with physician oversight — the highest-quality sourcing channel available.
Alternatives and comparisons
BPC-157 vs. TB-500
Both peptides target tissue repair, but through different mechanisms and scopes. BPC-157 acts locally through VEGFR2 angiogenesis and NO modulation. TB-500 acts systemically through actin regulation and cell migration. Both are WADA-prohibited under S0 and classified as FDA Category 2. See the comparison table in the stacking section above.
BPC-157 vs. Pentadeca Arginate (PDA)
PDA is not a different peptide. It is the arginate salt form of the identical BPC-157 amino acid sequence. The arginate counter-ion is designed for improved oral bioavailability and pH stability. Arginate is preferred for oral or GI-targeted protocols; acetate is preferred for injection.
Non-peptide alternatives by condition
Musculoskeletal injury. Platelet-Rich Plasma (PRP) injections carry stronger human clinical evidence and potential insurance coverage for some conditions. Eccentric loading exercises represent the strongest evidence-based intervention for tendinopathy.
Joint support. Collagen supplementation and omega-3 fatty acids are available over the counter with established safety profiles.
Gut healing. L-glutamine has well-established evidence for intestinal barrier support. Standard IBD medications (5-ASA compounds, biologics) carry FDA approval.
General recovery. Physical therapy and progressive resistance training remain the most evidence-supported recovery interventions.
Non-peptide alternatives generally carry stronger human clinical evidence, established safety profiles, and clearer legal status compared to BPC-157.
Ready to learn whether BPC-157 fits your health goals? Consult a licensed physician to discuss whether peptide therapy is appropriate for your specific situation.
Frequently asked questions
What is BPC-157?
BPC-157 is a synthetic pentadecapeptide — 15 amino acids, 1,419 Da molecular weight — derived from a protein in human gastric juice. Dr. Predrag Sikiric at the University of Zagreb identified it in 1993. The peptide promotes tissue repair through VEGFR2-mediated angiogenesis, nitric oxide modulation, and FAK-paxillin cell migration signaling.
Does BPC-157 cause cancer?
BPC-157 upregulates VEGFR2 (blood vessel growth) and FAK-paxillin (cell migration), both of which play roles in tumor development and metastasis. No carcinogenic effects have been documented in any published study. The EGR-1/NAB2 feedback loop may self-limit excessive angiogenic signaling, and one in vitro study (Radeljak et al., 2004) showed reduced VEGF signaling in melanoma cells. No dedicated in vivo tumor biology study has been conducted. BPC-157 is contraindicated for individuals with active cancer or cancer history.
How long does BPC-157 take to work?
BPC-157’s pharmacological half-life is under 30 minutes. Users commonly report initial effects — pain reduction, improved digestion — within 5–10 days of consistent dosing. Tissue-level healing responses typically require 2–8 weeks depending on injury type and severity. No controlled trials have measured time-to-effect in humans.
Can athletes use BPC-157?
No. WADA prohibits BPC-157 under S0 (Non-Approved Substances) since 2022, banning it at all times, in and out of competition, with no Therapeutic Use Exemption available. The NCAA, NFL, UFC, NBA, NHL, MLB, and DoD/OPSS also prohibit BPC-157. Advanced mass spectrometry detects BPC-157 in urine for up to 4 days.
What is the difference between BPC-157 acetate and arginate salt forms?
Both forms contain the identical 15-amino acid peptide sequence. The acetate salt is the standard injectable form with approximately 3% oral bioavailability. The arginate salt (PDA) bonds the peptide to L-arginine for improved pH stability, with patent-claimed oral bioavailability up to approximately 90% — based on one rat study, with no human confirmation. Arginate is preferred for oral or GI-targeted protocols; acetate is preferred for injection.
Does BPC-157 show up on drug tests?
Standard workplace drug panels do not test for BPC-157. WADA, USADA, NCAA, and military anti-doping tests can detect BPC-157 via high-resolution mass spectrometry. The Lee and Burgess (2025) pilot study showed plasma levels return to baseline within 24 hours after IV infusion, but urine detection extends up to approximately 4 days.
Are there any completed human clinical trials for BPC-157?
Three published human studies exist as of early 2026 — all from the same research group. Lee and Padgett (2021) studied intra-articular knee injections in 16 patients. Lee et al. (2024) studied intravesical bladder injections in 12 interstitial cystitis patients. Lee and Burgess (2025) studied IV safety in 2 healthy adults. A Phase I trial (NCT02637284) enrolled 42 volunteers in 2015 and was cancelled in 2016 with no results published. Zero completed randomized controlled trials exist.
Is BPC-157 the same as Pentadeca Arginate (PDA)?
Yes. PDA is the arginate salt form of the identical BPC-157 peptide sequence — same amino acids, different counter-ion formulation designed for improved oral stability and bioavailability. Diagen holds the patent for the arginate formulation.
References
- Sikiric P, Petek M, Seiwerth S, et al. Pentadecapeptide BPC 157 attenuates gastric lesions induced by restraint stress, cysteamine and ethanol in rats. Life Sciences. 1993;54:PL63-68.
- Sebecic B, Nikolic V, Sikiric P, et al. Osteogenic effect of pentadecapeptide BPC 157 on bone repair in rats. Journal of Orthopaedic Research. 1999;17(3):350-355.
- Staresinic M, Sebecic B, Patrlj L, et al. Gastric pentadecapeptide BPC 157 accelerates healing of transected rat Achilles tendon. Journal of Orthopaedic Research. 2003;21(6):976-983.
- Krivic A, Anic T, Seiwerth S, Huljev D, Sikiric P. Achilles detachment in rat and stable gastric pentadecapeptide BPC 157: promoted tendon-to-bone healing and opposed corticosteroid aggravation. Journal of Orthopaedic Research. 2006;24(5):982-989.
- Cerovecki T, Bojanic I, Brcic L, et al. Pentadecapeptide BPC 157 (PL 14736) improves ligament healing in the rat. Journal of Orthopaedic Research. 2010;28:1155-1161.
- Tudor M, Jandric I, Marovic A, et al. Traumatic brain injury in mice and pentadecapeptide BPC 157 effect. Regulatory Peptides. 2010;160(1-3):26-32.
- Chang CH, Tsai WC, Hsu YH, Pang JH. Pentadecapeptide BPC 157 enhances growth hormone receptor expression in tendon fibroblasts. Molecules. 2014;19:19066-19077.
- Vukojevic J, Milavic M, Perovic D, et al. Pentadecapeptide BPC 157 and the central nervous system. Neural Regeneration Research. 2022;17(3):482-487.
- He L, Feng D, Guo H, et al. Pharmacokinetics, distribution, metabolism, and excretion of body-protective compound-157 in rats and dogs. Frontiers in Pharmacology. 2022;13:1026182.
- Xu C, Sun L, Ren F, et al. Preclinical safety evaluation of body protective compound-157, a potential drug for treating various wounds. Regulatory Toxicology and Pharmacology. 2020;114:104665.
- Lee E, Padgett B. Intra-articular injection of BPC 157 for multiple types of knee pain. Alternative Therapies in Health and Medicine. 2021;27(4):8-13.
- Lee E, Walker C, Ayadi B. Effect of BPC-157 on symptoms in patients with interstitial cystitis: a pilot study. Alternative Therapies in Health and Medicine. 2024;30(10):12-17.
- Lee E, Burgess K. Safety of intravenous infusion of BPC157 in humans: a pilot study. Alternative Therapies in Health and Medicine. 2025;31(5):20-24.
- Vasireddi N, Hahamyan H, Salata MJ, et al. Emerging use of BPC-157 in orthopaedic sports medicine: a systematic review. HSS Journal. 2025. doi:10.1177/15563316251355551.
- McGuire FP, Martinez R, Lenz A, Skinner L, Cushman DM. Regeneration or risk? A narrative review of BPC-157 for musculoskeletal healing. Current Reviews in Musculoskeletal Medicine. 2025;18(12):611-619.
- Jozwiak M, Bauer M, Kamysz W, Kleczkowska P. Multifunctionality and possible medical application of the BPC 157 peptide: literature and patent review. Pharmaceuticals. 2025;18(2):185.
- Sikiric P, Seiwerth S, Skrtic A, et al. BPC 157 therapy: targeting angiogenesis and nitric oxide. Pharmaceuticals.2025;18(10):1450.
- Seiwerth S, Milavic M, Vukojevic J, et al. Stable gastric pentadecapeptide BPC 157 and wound healing. Frontiers in Pharmacology. 2021;12:627533.
- Gwyer D, Wragg NM, Wilson SL. Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing. Cell and Tissue Research. 2019;377(2):153-159.
Disclaimer: This content is for educational purposes only and does not constitute medical advice. BPC-157 is not approved by the FDA for any therapeutic indication. No peptide discussed in this article should be used without the supervision of a licensed healthcare provider. PeptideRx does not sell peptides or provide medical consultations. All dosing information reflects reported clinical and anecdotal ranges, not FDA-approved protocols. Consult your physician before beginning any peptide protocol. Always verify product quality through independent third-party testing. If you are a competitive athlete, military service member, or government employee, BPC-157 may be prohibited by your governing organization.